  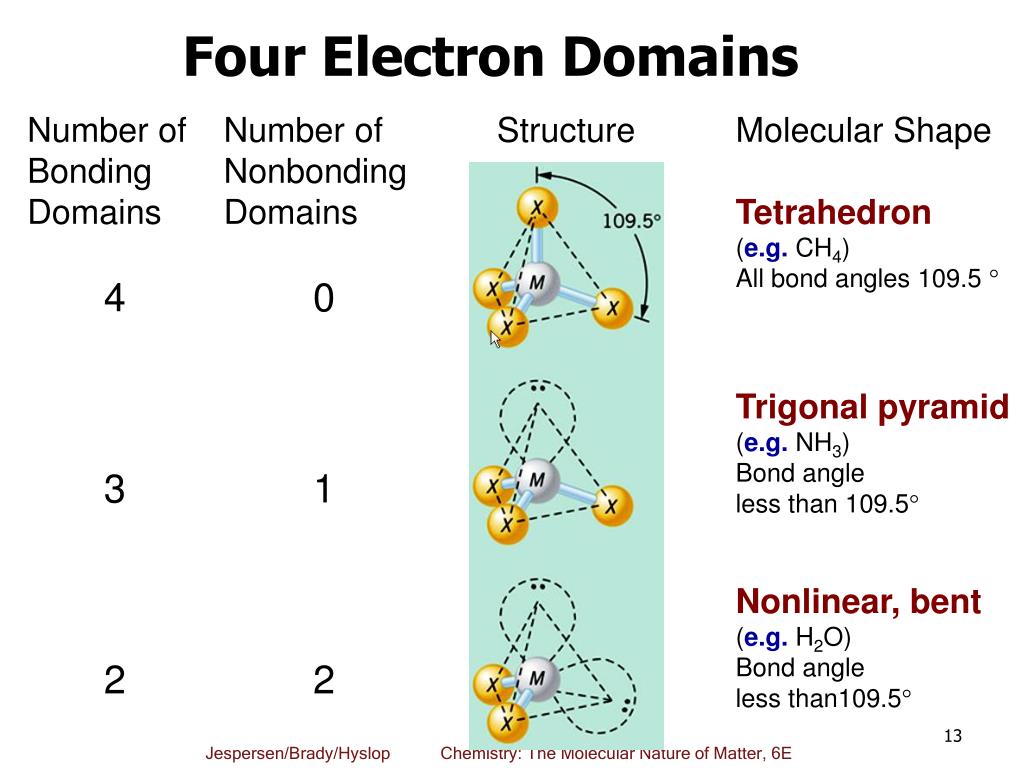
With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. Choose a central atom: Both the atoms will be central. Look for the total number of bonds forming: Double covalent bonds are forming in an O2 molecule. Find how many electrons are needed: It is four for one O2 molecule. There are a total of 4 electron density regions around the central S atom in the Lewis structure of SO32-. Find total valence electrons: It is two for each oxygen atom. The sulfur (S) is present at the center of the molecular ion while oxygen (O) occupies the terminals, one on each side. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. The Lewis structure of sulfite SO32- ion is made up of a sulfur (S) atom and three oxygen (O) atoms. The term electron-pair geometry is the name of the geometry of the electron-pair/groups/domains on the central atom, whether they are bonding or non-bonding. Pi-bonds are absent, making the structure. 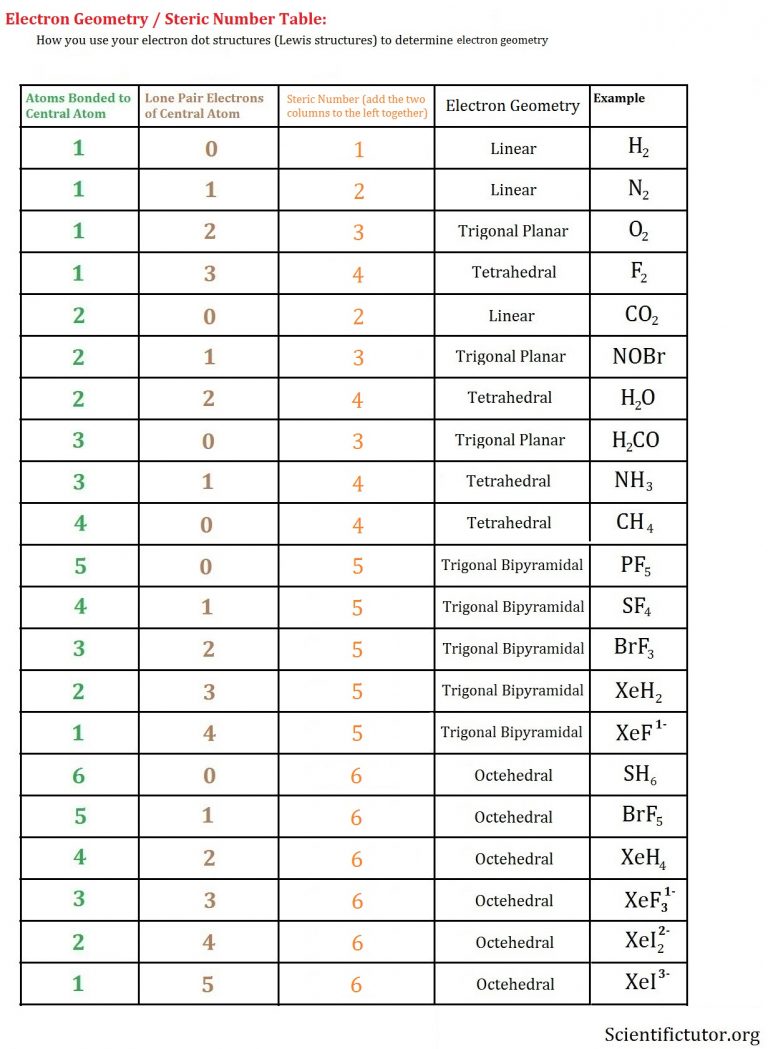
We calculate the number of total electron pairs in electron geometry and not in molecular geometry. The NH 4+ ion is overall non-polar (net µ 0) due to its symmetrical shape and geometry. The NH 4 + ion has an identical electron geometry and molecular geometry or shape i.e., tetrahedral. Sigma bonds are formed between Carbon and Fluorine. In molecular geometry, though, we only consider bond electron pairs. The positive 1 charge present on the ion accounts for 1 valence electron removed in its Lewis structure. To know more about the hybridization of CF 4 in detail, we look into the nature of the C-F bond. This is especially true for non-polymeric arrangements. The electron geometry for BrO3- is tetrahedral. Hence, the molecular shape or geometry for BrO3- is trigonal pyramidal. We designate SF 4 as AX 4E it has a total of five electron pairs. The number of electron domains is an easy way to determine the hybridization of a molecule. So, as per the VSEPR chart, if the central atom of a molecule contains 1 lone pair and is covered by three surrounding atoms, then the molecular shape of that molecule is trigonal pyramidal in nature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
